Cannabinoid Knowledge
Over the years, the term ‘cannabinoids’ has experienced a shift in meaning.
What are Cannabinoids?
Whereas in the past only the naturally occurring substances in the hemp plant (phytocannabinoids) were referred to as cannabinoids, today messenger substances found in the human body (endocannabinoids) as well as artificially produced compounds (synthetic and semi-synthetic cannabinoids) are also included in this group. Although endocannabinoids and synthetic compounds often differ significantly in chemical structure from plant cannabinoids such as Δ9-tetrahydrocannabinol (THC) and cannabidiol (CBD), they also interact with the so-called endocannabinoid system (ECS) of the human body, resulting in a wide range of pharmacological effects.
In the following, we will primarily take a closer look at the cannabinoids of the hemp plant:
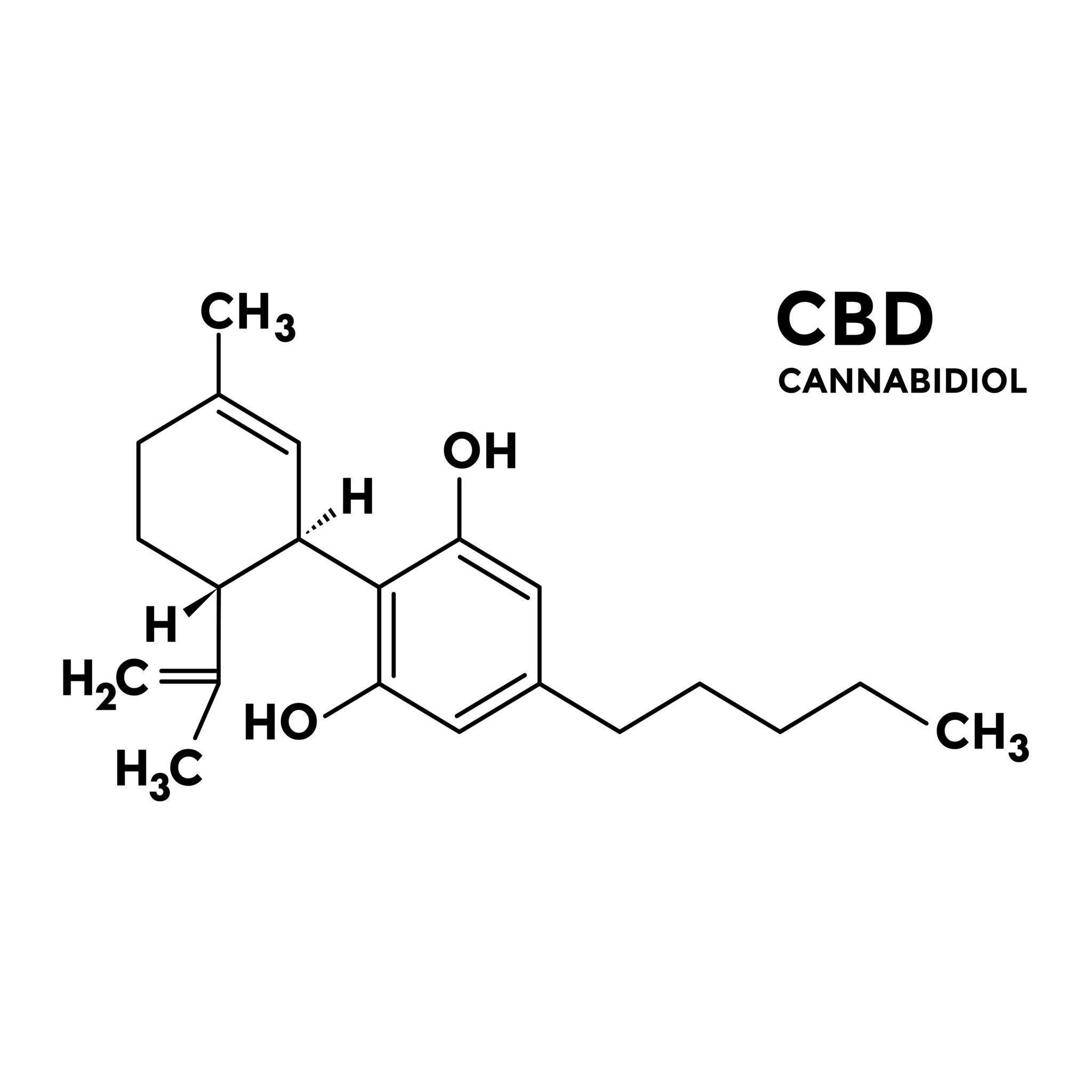
Phytocannabinoids – Cannabinoids of the Hemp Plant
Phytocannabinoids are a group of substances currently comprising more than 120 different compounds. Chemically, they are so-called terpenophenols, produced and stored in the glandular hairs (trichomes) of the hemp plant. Since the inflorescences of female plants are densely covered with these glandular hairs, they exhibit the highest cannabinoid concentrations. It is assumed that these substances serve the plants as a defense against herbivores and harmful microorganisms (bacteria and fungi).
In addition to the typically predominant and better-known representatives such as THC and CBD, there is a wide variety of other phytocannabinoids. Although often present only in trace amounts, they modify the effects of the main components and thus play a decisive role in shaping the overall pharmacological profile of each cannabis strain.
We offer quantitative determination of the 11 most important phytocannabinoids as a complete package!
What is the Difference Between THC and THCA or CBD and CBDA?
Cannabinoids marked with the suffix A (for “acid”) are so-called acidic cannabinoids, which are the precursors to the more commonly known neutral cannabinoids. These substances, also referred to as cannabinoid carboxylic acids (or simply cannabinoid acids), are the original components produced by the plant and make up the majority of the cannabinoid fraction in, for example, fresh flowers.
Over time, cannabinoid acids are converted into their neutral forms by releasing carbon dioxide; for example, THCA degrades to THC and CBDA to CBD.
This process, called decarboxylation, usually occurs gradually but can be greatly accelerated by heat exposure. Thus, in cannabis extracts produced with heat, the original acidic forms are often present only in trace amounts. Likewise, when smoking cannabis products, these acids are immediately and completely converted into their neutral analogs.
Although the neutral cannabinoids generally have much stronger effects than the acidic forms, the acidic cannabinoids also exhibit unique effects. CBDA and CBGA, in particular, continue to attract scientific interest due to their anti-inflammatory and antiviral properties.
As part of our quantitative cannabinoid analysis, acidic and neutral cannabinoids are measured both separately and in total. For example, the designation “Total THC” represents the sum of acidic and neutral THC in your sample material.
How Does the Biosynthesis of Cannabinoids in the Hemp Plant Occur?
The starting point for most other cannabinoids in the plant is cannabigerolic acid (CBGA), which is converted into other cannabinoid acids by specific enzymes, the so-called synthases. For example, THCA synthase catalyzes the conversion of CBGA into THCA, while CBDA synthase and CBCA synthase are responsible for the formation of CBDA and CBCA, respectively.
As mentioned in the previous section, these cannabinoid acids can be converted into their neutral forms through decarboxylation. This process occurs non-enzymatically and can therefore also take place outside the living plant, as is the case during storage of dried flowers. A similar phenomenon occurs with cannabinol (CBN), which forms long after harvest through oxidative degradation of THC.
Through targeted artificial selection and crossbreeding of cannabis plants with favored traits, it has been possible in recent decades to generate a variety of new genetics that differ drastically in their cannabinoid profiles (so-called chemotypes). The molecular cause for this can be found in the plant’s genes: for example, in almost THC-free CBD hemp varieties, the gene encoding THCA synthase is defective, so the enzyme is no longer produced properly. In THC-rich strains, by contrast, the CBDA synthase is missing, shifting the ratio in favor of psychoactive THC.
What are Synthetic and Semi-Synthetic Cannabinoids?
The term “synthetic” generally means that a substance is not obtained from natural resources but produced artificially in a chemical laboratory. Thus, naturally occurring cannabinoids such as CBD and Δ9-THC can not only be extracted from the hemp plant but also synthesized, i.e., chemically recreated. The resulting “nature-identical” cannabinoids do not differ in any way—chemically or pharmacologically—from their natural counterparts.
In the narrower sense, however, synthetic cannabinoids primarily refer to substances that exhibit pharmacological effects similar to phytocannabinoids but are neither chemically related nor naturally occurring. An example is the cannabinoid JWH-018, which can only be produced synthetically, was legally available for many years in the form of herbal mixtures (“Spice”), and became infamous due to several fatalities.
Semi-synthetic cannabinoids are also produced in the laboratory. In contrast to fully synthetic cannabinoids like JWH-018, these always start from naturally occurring cannabinoids such as CBD and THC, which are then chemically modified. In semi-synthetic cannabinoids, the basic phytocannabinoid structure is usually still recognizable, showing their chemical relationship to this group. An example is hexahydrocannabinol (HHC), a psychoactive cannabinoid that occurs naturally only in trace amounts but can be easily synthesized by hydrogenating THC.
What is the Human Endocannabinoid System (ECS)?
The fact that cannabinoids from the hemp plant trigger various pharmacological effects in humans led early on to the assumption that the human body must also produce its own messenger substances (endogenous cannabinoids or endocannabinoids) with similar effects. Furthermore, there must also be receptors through which these messenger substances exert their specific actions.
Indeed, anandamide (N-arachidonoylethanolamide, AEA) and related substances were later identified as endogenous ligands, along with two subtypes of cannabinoid receptors (CB1 and CB2). The interplay of these biochemical factors is referred to as the human endocannabinoid system.
CB1 receptors are mainly located in the nervous system and are involved in regulating physiological processes such as pain transmission, hunger, and sleep induction. CB2 receptors, found in bone tissue and immune cells, influence important functions of the human immune system.
When plant-based or synthetic cannabinoids are introduced into the body, endogenous regulatory circuits can be influenced in various ways. For example, the intoxicating effect of THC is primarily mediated via CB1 receptors in nerve cells, while non-psychoactive CBD exerts its immunomodulatory effects mainly through CB2 receptors. Beyond modulation of the ECS, the complex pharmacological profile of cannabinoids results from numerous interactions with other receptor systems in the human body.